Trial Results Support At-Home Treatment of Edema in Heart Failure with Subcutaneous Furosemide as an Effective and Safe Alternative to Inpatient Hospital Care
PR Newswire
BURLINGTON, Mass., May 11, 2026
SUBCUT HF II, a randomized controlled investigator-sponsored trial of at-home subcutaneous furosemide for heart failure–related edema, meets its primary and key secondary endpoints, supporting the safety and efficacy of patient self-treatment outside the hospital.
BURLINGTON, Mass., May 11, 2026 /PRNewswire/ -- SQ Innovation today announced topline results from the SUBCUT HF II trial of its product Lasix® ONYU, a novel subcutaneous formulation of furosemide for people with heart failure-associated edema. Patients randomized to treatment at home spent an average of four (4) fewer days in the hospital compared to those treated with standard-of-care intravenous (IV) furosemide, according to the trial's results, presented May 9 at Heart Failure 2026, the annual congress of the Heart Failure Association (HFA) of the European Society of Cardiology (ESC), in Barcelona, Spain.
SUBCUT HF II is a multicenter, open-label, randomized controlled trial conducted across 20 National Health Service (NHS) hospitals in the U.K. The trial was co-sponsored by the University of Glasgow and the National Health Service (U.K.) Greater Glasgow and Clyde Health Board and led by Principal Investigators Dr. Ross T. Campbell and Professor Mark C. Petrie of the Glasgow Cardiovascular Research Centre, University of Glasgow.
"Edema in heart failure is a leading reason for hospital admissions worldwide," said Professor Petrie. "This novel treatment option relies on the same therapeutic fundamentals that have been in place in the hospital since the 1960s, and enabling this to now be done at home by a patient or caregiver fundamentally changes the nature and trajectory of this very common and burdensome disorder."
The trial evaluated at-home treatment of heart failure patients as an alternative to standard hospital treatment. For the study, 172 patients with heart failure were enrolled shortly after they were admitted to a hospital for treatment of edema or fluid overload. Ninety-two patients were randomized to receive at-home treatment with Lasix ONYU and 80 received in-hospital treatment with IV diuretics.
Patients randomized in the study had a mean age of 71 years and were representative of a typical hospitalized heart failure population requiring diuretic therapy, with a median NT-proBNP of 3,365 pg/mL, peripheral edema present in 92%, and pleural effusion in 29%.
The primary endpoint was an avoidance of a minimum of 3.5 days of inpatient care also referred to as Days Alive and Out of Hospital (DAOH) over the 30-day period starting with the moment of randomization. The primary endpoint reflects that the at-home therapy was effective and safe with low readmission due to treatment failure or admission for complications.
The study demonstrated four (4) more days on its primary endpoint of DAOH at 30 days (p<0.001).
Secondary endpoints included:
- Length of index hospitalization,
- Change in health status at 60 days (assessed by the KCCQ-12),
- Days spent alive and out of hospital from randomization to 60 days
- Total number of HF hospitalizations at 60 days
- CV death or first HF hospitalization at 60 days
- CV death at 60 days
- Safety as determined by treatment emergent adverse events (TEAEs)
- Device failures
The researchers reported length of index hospitalization reduced by 5.5 days (p<0.001), and benefits of DAOH remained at 60 days (4.23 DAOH difference between groups – p<0.001).
Secondary endpoints supported that aside from the avoidance of inpatient days, at-home Lasix ONYU treatment was equivalent to in-hospital treatment.
Patients in the at-home treatment arm received a full therapeutic dose by self- administration of approximately 11 units on average (880 mg). This total dose for an episode of edema in heart failure is consistent with published literature for in-hospital treatment.
Adverse events were infrequent, and no device-related serious adverse event occurred. Lasix ONYU treatment was generally well tolerated.
"The SUBCUT HF II study represents an important milestone on this path home for the many patients for whom this is an option," said Pieter Muntendam, MD founder, President and CEO of SQ Innovation, the manufacturer of Lasix ONYU. "This is the first outcomes study of home use of subcutaneous furosemide that meets the goals of demonstrating hospital avoidance and safe home use."
Edema (fluid overload) is a leading cause of hospitalizations globally for people over 65 years old with heart failure, and typically requires inpatient IV diuretic therapy that could take up to one week. In the U.S., over 1.2 million people are admitted for heart failure each year, significantly contributing to Medicare and other health insurance expenses amid ongoing hospital overcrowding.
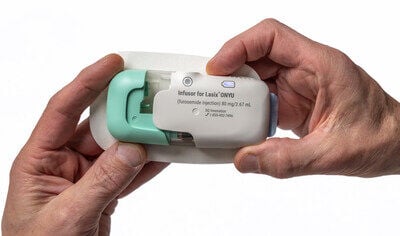
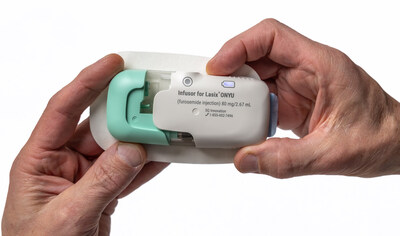
Lasix ONYU is a novel drug-device combination that was approved by the U.S. Food and Drug Administration (FDA) in October 2025, for the treatment of edema in adult patients with chronic heart failure. The Lasix ONYU Infusor slowly administers 80 mg of the diuretic furosemide over a period of five hours, resulting in significant diuresis similar to IV, but in a controlled manner designed for at-home administration to minimize risk of orthostatic hypotension and falls.
About SUBCUT HF II
SUBCUT HF II (ClinicalTrials.gov: NCT05419115) is a multicenter, prospective, open-label, parallel-group, active comparator, phase II randomized controlled trial evaluating the safety and efficacy of an early supported discharge strategy using a novel subcutaneous formulation of furosemide in patients hospitalized due to heart failure. Full details of the trial design have been published in the European Journal of Heart Failure (doi: 10.1093/ejhf/xuaf018).
About SQ Innovation
SQ Innovation, Inc. is a privately held Swiss biopharmaceutical company with offices in Zug, Switzerland, Burlington, MA, USA, and Rotterdam, The Netherlands. The company was founded to develop and commercialize innovative, cost-effective therapies for subcutaneous delivery, enabling at-home treatment for conditions that are usually managed during hospitalizations. SQ Innovation has developed a novel drug-device combination for treating edema in adult patients with chronic heart failure — a condition typically requiring intravenous administration of diuretics in a hospital setting. Lasix ONYU was developed with consideration for patients, payors, healthcare providers, and environmental impact. Lasix ONYU received approval from the U.S. Food and Drug Administration on October 7, 2025. For more information about Lasix ONYU, including important safety information and the full prescribing information, please visit www.lasix-onyu.com.
About Lasix ONYU
Lasix® ONYU is a drug-device combination that was approved by the U.S. Food and Drug Administration on October 7, 2025, for the treatment of edema in adult patients with chronic heart failure. Lasix ONYU is available for ordering from Cardinal and Cencora and for dispensing and overnight delivery from Walmart Specialty Pharmacy. The pharmaceutical component of Lasix ONYU is a novel, high-concentration formulation of the diuretic furosemide, at 30 mg/mL. It comes in a pre-filled glass cartridge containing 80 mg of furosemide in 2.67 mL. The Lasix ONYU Infusor consists of two main parts: the Reusable Unit and the Disposable Unit. The Reusable Unit is an electromechanical device that contains the battery, motor, and electronic components necessary for operation and safety functions. It can be used up to 48 times before it can be recycled. The Disposable Unit is a sterile, single-use plastic component that holds the drug cartridge. It includes a micro-`piston pump, fluid path, needle insertion and retraction mechanism, and a 29-gauge needle. After placement on the abdomen, the needle penetrates the skin when the device is activated. The Lasix ONYU Infusor slowly administers 80 mg furosemide over a period of five hours. This method results in significant diuresis similar to IV, but in a more controlled manner. This avoids the brief, intense diuretic effect that occurs with rapid IV infusion or injection. Lasix ONYU is a second-generation combination product for subcutaneous furosemide that follows the introduction of Furoscix in 2023. The advanced two component design of Lasix ONYU offers benefits for patients, healthcare providers, payers, and the environment. For Important Safety Information, Prescribing Information and Instructions for Use, visit www.lasix-onyu.com.
INDICATION
Lasix® ONYU is a loop diuretic indicated for the treatment of edema in adult patients with chronic heart failure.
IMPORTANT SAFETY INFORMATION
Before using Lasix® ONYU, read the Instructions for Use and tell your healthcare provider about all your medical conditions, including if you are allergic to furosemide or any of the ingredients in Lasix ONYU, have trouble urinating, or if you are allergic to medical adhesives.
Tell your healthcare provider about all of the medicines you take, including prescription and over-the-counter medicines.
Warning: Only use the Lasix ONYU Prefilled Cartridge with the Lasix ONYU Infusor. Do not use insulin cartridges or other medicine cartridges in the Lasix ONYU Infusor. Doing so could cause severe injury.
What are the possible side effects of Lasix ONYU?
- Dehydration: Lasix ONYU is a diuretic that can make you lose a lot of fluid and with it electrolytes. You may get a dry mouth, have increased thirst, get muscle pains or cramps, have reduced urine output or your urine may be more yellow than normal, you may get a headache, get dry skin, or have nausea or vomiting. Your healthcare provider may check your electrolytes while receiving Lasix ONYU.
- Low Blood Pressure: Lasix ONYU may lower your blood pressure temporarily. You may feel lightheaded or dizzy. This usually happens when you stand. Getting up slowly may help.
- High Blood Sugar: Lasix ONYU may increase blood sugar (glucose) levels if you have diabetes mellitus.
- Loss of Hearing: Lasix ONYU can cause ringing in your ears. If so, tell your healthcare provider.
- Risk of Sunburn: Your skin may be more sensitive to sunlight while taking Lasix ONYU.
- Infusion Site Reactions: Lasix ONYU can cause infusion site pain, bruising and temporary swelling or redness at the site of the Infusor.
- Incomplete Dosing: Make sure the Infusor does not get wet during use. Also limit your physical activities. Some movements or when it gets wet may stop the infusion and you may not get all the medication.
These are not all the possible side effects of Lasix ONYU. For more information, ask your healthcare provider or pharmacist.
Call your healthcare provider for medical advice about side effects. You may report side effects to the FDA at 1-800-FDA-1088.
Please see the full Prescribing Information and Instructions for Use at www.lasix-onyu.com.
Cautionary Note Regarding Forward-Looking Statements
This press release contains statements that may or are considered "forward-looking statements. All statements other than statements of historical facts are "forward-looking statements," including those relating to future events. In some cases, you can identify forward-looking statements by terminology such as "anticipate," "believe," "continue," "expect," "may," "on track," "plan," "potential," "target," "will," or "would", the negative of terms like these, or other words or terms of similar meaning. These may include implied or express statements about our expectations regarding strategy, business plans, and objectives on the development of Lasix ONYU; decisions by regulatory authorities regarding marketing approval or material restrictions, limitations, or obligations on marketing and market access of the Company's products; delays in arranging satisfactory manufacturing capabilities and establishing commercial infrastructure; the risk that third party payors (including governmental agencies) will not reimburse for the use of products at acceptable rates or at all; the risk that third parties will not agree to license any necessary intellectual property on reasonable terms or at all; and other risks set forth from time to time. Any forward-looking statements in this press release are based on management's current expectations and beliefs and are subject to a number of risks, uncertainties, and important factors that may cause actual events or results to differ materially from any forward-looking statements contained in this press release. In addition, any forward-looking statements represent our views only as of today and should not be relied upon as representing our views as of any subsequent date. We explicitly disclaim any obligation to update any forward-looking statements. No representations or warranties (expressed or implied) are made about the accuracy of any such forward-looking statements.
Notes: Lasix® ONYU is the proprietary name for the furosemide injection combination product in the US. The trademark Lasix® is registered for Validus Pharmaceuticals L.L.C. in the United States and used by SQ Innovation under license. The proprietary name for Europe has not yet been established. Furoscix® is a registered trademark of scPharmaceuticals, Inc.
For Media Inquiries:
pr-media@sqinnovation.com
Ryan Jaslow; ryan@jaslowhealthcomms.com
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/trial-results-support-at-home-treatment-of-edema-in-heart-failure-with-subcutaneous-furosemide-as-an-effective-and-safe-alternative-to-inpatient-hospital-care-302768025.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/trial-results-support-at-home-treatment-of-edema-in-heart-failure-with-subcutaneous-furosemide-as-an-effective-and-safe-alternative-to-inpatient-hospital-care-302768025.html
SOURCE SQ Innovation